 
Stays current with the ongoing changes in the pharmaceutical regulatory environment, i.e.Conforms to training schedule for own position and maintains awareness of SOPs contents according to company requirements.Preparation and revision of related SOPs.Responsible for updating the protocol related concerns to investigator(s), sponsor and IEC.Approval of protocols from Ethics committee in consultation with Principal Investigator and Sponsor.Coordination with study personnel and other departments for protocol related issues for finalization of protocol. 
Preparation of Protocols, Informed Consent Forms, Case Report Forms, and other protocol related documents as per applicable regulatory requirements in consultation with Group In-charge, Principal Investigator, Biostatistician, Analytical Investigator, and Head of the department.Cliantha has fifteen years of impeccable regulatory history with USFDA, WHO, MHRA, Health Canada, AGES, AEMPS, MCC, MOH, ANSM, MOPH, ANVISA, CAP, and NABL.īasic qualification required : B.Pharm / M. 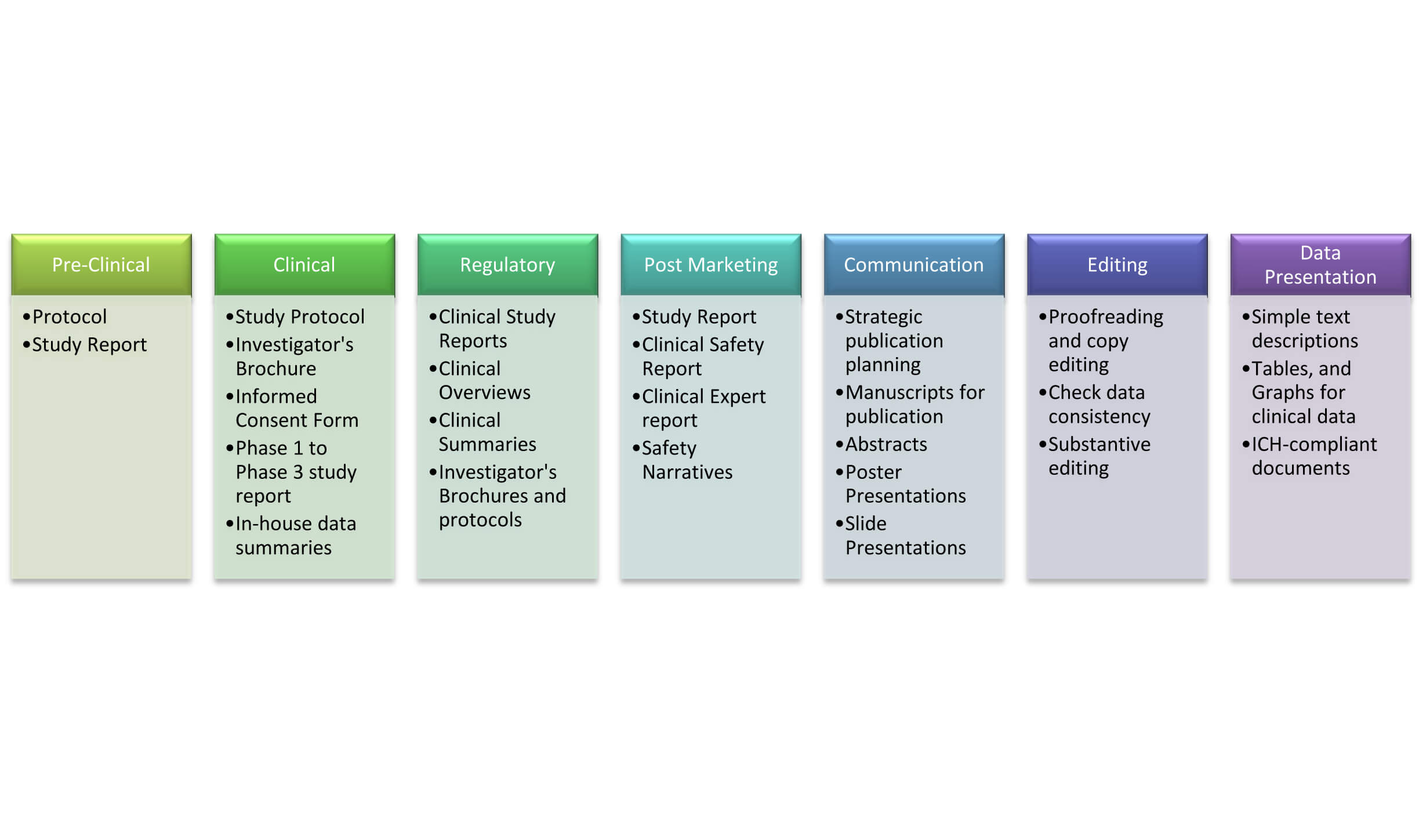
Cliantha’s motto is Science with Integrity. Basic Functioning working knowledge of software programs in Windows environment.Cliantha Research, a full-service Clinical Research Organization (CRO), is a leading provider of Clinical research services, based in Ahmedabad, India. Experience in working with collaborative, cross-functional teams. Your technical expertise as a Lab Tech II in the area of either Quality Control/Quality Assurance functions will contribute to our client's innovative therapies which will impact the quality and duration of life.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
